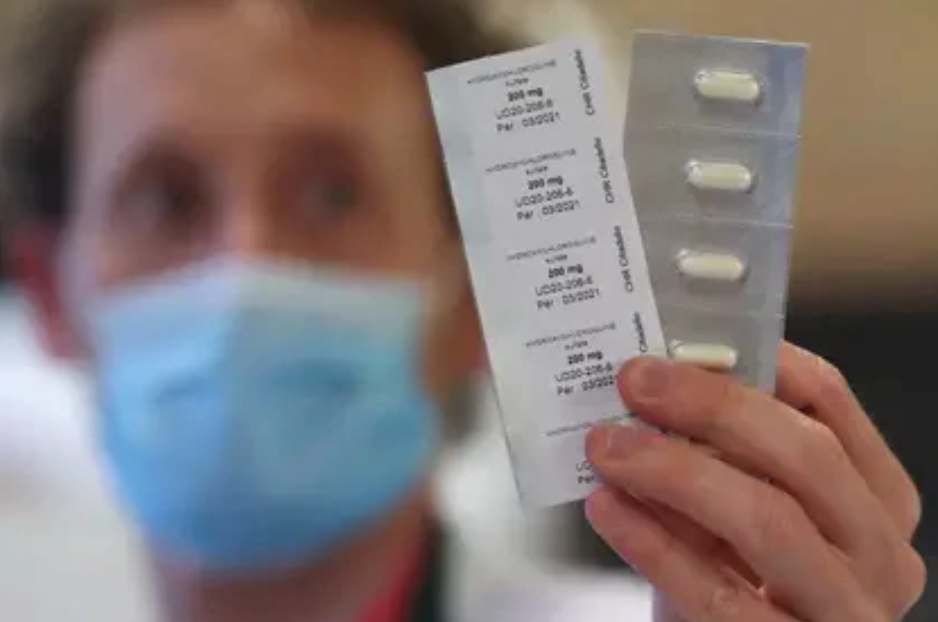
In its fight to contain the health crisis of the coronavirus in Mexico, the Ministry of Health will supply hydroxychloroquine to 20,000 outpatients of coronavirus starting next week.
It is a treatment to combat other autoimmune diseases, and the Federal Commission for the Protection against Health Risks (Cofepris) has already authorized the Ssa to administer the drug.
The drug will be offered to patients who have the virus, despite the fact that various scientists have warned that it increases the risk of arrhythmias and death and shows no benefit to COVID-19.
The hydroxychloroquine will be supplied in a controlled manner and out of clinical trials, revealed José Sifuentes, director of the National Institute of Medical Sciences and Nutrition Salvador Zubirán after receiving a donation of 130,000 doses of the generic drug from Novartis.
Two months ago, the Swiss pharmaceutical company Novartis disclosed the donation of doses of hydroxychloroquine, a drug that is used to treat diseases such as malaria or lupus.
“What we have agreed with the Secretary of Health, Jorge Alcocer, is to distribute it to national institutes of health, regional hospitals, of high specialty and with institutions of social security”, clarified Sifuentes.
Mexico has 62,527 confirmed cases of COVID-19, and there are 6,989 deaths as of Saturday, April 23. However, Mexico reports there are only 13,300 active cases of the pandemic nationally, and continues to suggest that the pandemic curve has flattened in the country, although cases continue to climb each day.
The World Health Organization (WHO) warned that the population should not take the drug hydroxychloroquine to combat COVID-19.
Mike Ryan, WHO executive director for Health Emergencies, said the drug should only be used in clinically monitored clinical studies,” due to its potential side effects.”
In addition, a study published in The Lancet, found that the drug does not benefit patients with COVID-19, but was related to increased mortality rates and increased cardiac arrhythmias among hospitalized coronavirus patients.
This analysis, published in the Lancet journal, monitored the behavior of at least 15,000 patients with COVID, who were given the drug, in addition to data from 81,000 controlled patients.
According to Sifuentes, clinical trials carried out so far have shown that hydroxychloroquine has the benefit of reducing the generalized inflammation that occurs in infected people, so it will be used in patients with early stages of the disease.
He explained that the intention is to offer the medicine in a supervised and careful way, after “a secretarial agreement. This can be done, not only with face-to-face monitoring, but even with monitoring over the phone, or video calls”.
On Monday, May 18, the President of the United States, Donald Trump, surprised many after revealing that for weeks he consumed hydroxychloroquine and zinc preventively against the coronavirus, although he assured that he had no symptoms of the disease.
However, on Wednesday the 20th, Trump said he would stop taking hydroxychloroquine “in a day or two” after taking the drug for two weeks as prevention against COVID-19, a use that has not been scientifically proven or studied.
“I think the (drug) regimen ends in a day or two. I think it’s two days,” the president told reporters at the White House.
In its fight to contain the health crisis of the coronavirus in Mexico, the Ministry of Health will supply hydroxychloroquine to 20,000 outpatients . . .